 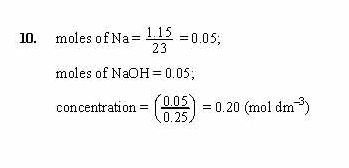
Without knowing the chemical or molecular formula of the compound and without knowing the atomic weights of the individual elements involved in the chemical formula it is very difficult to calculate the molar mass of the respective compound. The molar mass of the sodium hydroxide is 40 g/mol. Now we must express the molecular weight of the sodium hydroxide in molar mass. Molecular weight of the NaOH= 23 + 1 + (16) = 40. Molecular weight of the NaOH = Atomic weight of the Na + Atomic weight of the H + Where: Weight d Density (or specific gravity) MW Molecular Weight (or Formula Weight). Part B The most common source of copper (Cu) is the mineral chalcopyrite (CuFeS2). The following equation is used for calculating acid and base molarity where the concentration is given in wt : ( × d) / MW × 10 Molarity. What is the mass of 2.30 x 1022 formula units of NaOH (Molar mass 40.0 g/mol) Express the mass in grams to three significant figures. Consider the example given below 0.25 mol/L solution of sodium hydroxide means that 0.25 moles of sodium hydroxide have been dissolved in the 1 litre (or 1 cubic decimetre) of the solution. Now we can calculate the molar mass or molecular weight of the sodium hydroxide by using the formula below. Part D Sodium hydroxide, NaOH, is a strong base that is used in industrial synthesis and processes such as making paper. The standard unit of Molarity is moles/L, or Molar or M. The atomic weight of the sodium is 23, the atomic weight of the hydrogen is 1, and the atomic weight of the oxygen is 16. We should know the atomic weights of the sodium, hydrogen and the oxygen to calculate the molar mass of the sodium hydroxide. We should know the chemical formula of the sodium hydroxide to calculate the molar mass of it. In the question it is asked to calculate the molar mass of the sodium hydroxide. 
We should know the molecular or chemical formula of the respective compound. Hint: The molar mass of any compound can be calculated by adding the atomic weight of the individual atoms involved in the chemical formula of the respective chemical compound. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
